Female Stress Incontinence Procedures
JOIN OUR NEWSLETTER
Receive custom tools to help you manage your condition and get the latest in bladder and bowel health from NAFC!
FEMALE STRESS URINARY INCONTINENCE Procedures
Surgical treatment of stress urinary incontinence (SUI) has been the mainstay of therapy, particularly in the United States, for many years. While surgery is still generally acknowledged to be the most effective treatment for SUI, the myriad of operations attests to the fact that there is no one best operation for all patients.
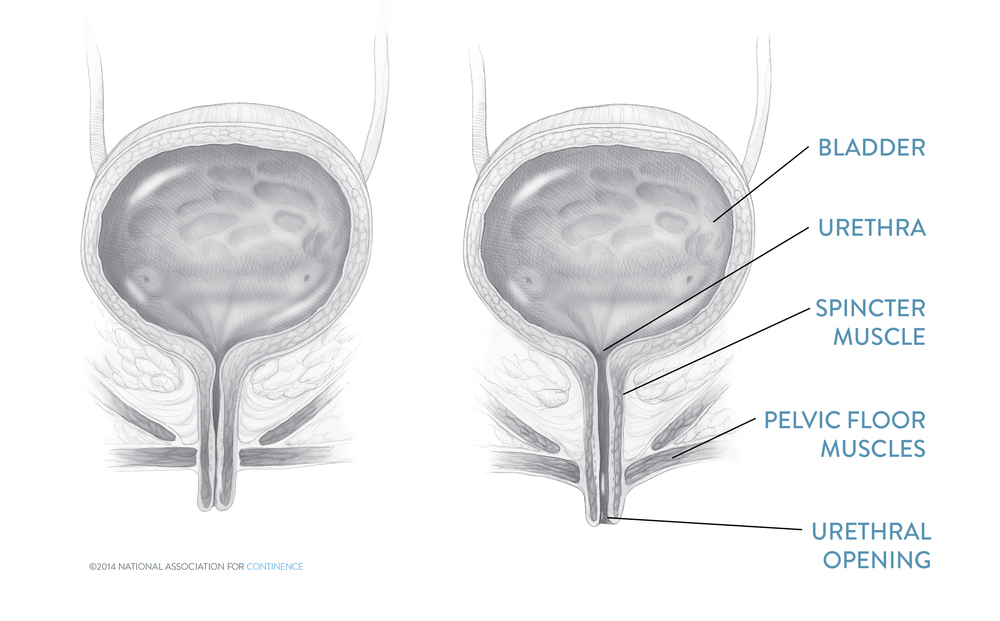
The vast majority of SUI procedures are now one of several different pubovaginal (i.e., in the region of the pubis and the vagina) slings. The basic concept of the sling is that a piece of strong material is placed beneath the urethra as a supporting “hammock.” The sling thus corrects the poor anatomic support of the urethra and may additionally provide a degree of compression to the urethra.
There are three main types or approaches of mid-urethral slings:
Retropubic
Transobturator
Mini-sling
All three are effective, but because the standard retropubic and transobturator slings enjoy 10 to 15 years of use and an impressive number of reports of their effectiveness, they are considered the gold standard for surgical treatment of SUI in women by many leading clinicians.
EVALUATION
When incontinence is severe enough to cause embarrassment or limits your activities, it is time to talk to your doctor. To determine which treatment is best for you, the doctor should take a detailed history of your general health and your bladder symptoms.
Click Here To Download A Free Bladder Diary
It is helpful for you to be prepared for this visit by keeping a bladder diary for a few days detailing the time of each urination and keeping track of the amount and circumstances of any urine leakage. It is very helpful if you can measure the volume of each urination for one full 24-hour period. This type of record can tell the doctor a great deal about your bladder function without invasive testing.
The doctor will want to know about all of your medical history, particularly details of childbirth and any pelvic surgery. As part of an evaluation for incontinence, it is important to consider other problems that may be related or that could be addressed at the same time. The doctor will ask you about bladder infections, difficulty urinating, gynecologic problems, problems with your bowels including fecal incontinence, and neurologic problems such as back injury, stroke, or other neurologic diseases.
If you have had prior treatment for incontinence—medical, surgical, or other therapies—the details of this will be important in making a decision about future treatment.
The doctor will then perform a physical examination and test your urine for infection or other problems. The doctor may catheterize you (i.e., pass a small tube through the urethra to drain the bladder) to determine if you are emptying the bladder completely.
The doctor may examine you while coughing and straining to see if stress incontinence can be demonstrated. This simple type of office evaluation is adequate for many patients, and treatment can often be started at this point.
For other patients, X-rays or MRI of the bladder, bladder function tests (urodynamic studies), which measure the bladder and urethral sphincter pressures, and cystoscopy, may be required. Cystoscopy is an examination that consists of looking inside of the bladder and urethra using a small telescope, usually performed in the doctor’s office.
SURGERY
Newer materials and a needle approach through the skin with minimal cutting, along with tension-free placement in the mid-urethra, have led to these operations becoming the most popular procedures in the United States and Europe. The mini-sling, utilizing the transobturator approach and a single incision, is generally placed with tension in the mid-urethra and is the newest among the three types.
There are now a number of competitive products offering slightly different sling material and/or methods of placement. While a popular and effective procedure, the route used to deliver the tension-free transvaginal tape does not allow surgeons a full view of the pelvic organs. One increasingly popular technique is the transobturator approach that uses a different incision site considered to lower the risk of urethral and bladder injury.
There are still other techniques that deliver synthetic slings, which are either meant to lower injury risk and/or achieve greater compression of the urethra. The tradeoff is greater risk of post-operative groin or pelvic pain with the transobturator approach. While mini-slings are considered as safe as the mid-urethral sling procedures performed today, the data are less mature and thus conclusiveness regarding equivalent effectiveness has not been established.
The mid-urethral slings are simple, well-tolerated procedures. The nylon-like mesh is just over a half inch in width and is inserted with needles under the mid-urethra.
After anesthesia, an opening is made under the mid-urethra large enough to admit one finger. In the lower pelvic area two small openings are made just to pass a needle. The sling is then threaded through this track. Because the material is a bit wider than the needles, it is adjusted loosely in the proper position and held in place with friction.
No stitches are needed to keep it in place. The vaginal opening is closed with a few stitches, and the needle exits are often closed with skin glue.
Suspension procedures can be performed through the vagina or through the abdomen. Although abdominal suspensions are less popular than slings, the Burch procedure is still considered to be a good choice for patients with SUI especially if it accompanies another abdominal procedure.
In the Burch procedure, an abdominal incision is made, the area where the urethra and bladder connect is surgically exposed and several (usually four) sutures of either permanent or absorbable material are placed next to the urethra at the bladder neck and mid-urethra and tied to a strong ligament nearby on the pelvic bone.
RECOVERY
Many surgeons perform the mid-urethral slings with the patient under sedation with local anesthesia. When only an incontinence procedure is performed, hospitalization is typically minimal—outpatient surgery or an overnight stay. If additional procedures are performed to correct pelvic organ prolapse, then hospitalization may be required. It is unusual for a patient to be in the hospital for more than two nights after a routine operation.
At the time of discharge, the patient should be able to walk without assistance, go up and down short flights of stairs, eat a regular diet, and manage the bladder. A catheter is rarely required after mid-urethral sling procedures, and most patients simply urinate normally, although the stream may be a bit slower.
Transient urinary retention (i.e., the inability to empty the bladder) may be expected for several days or up to two weeks as with other bladder neck suspension procedures.
The patient may have an indwelling urethral catheter for several days, may start intermittent catheterization (passing a small straw like tube into the bladder several times a day to empty), or a suprapubic tube (a small catheter exiting the bladder through the lower abdomen) may be placed during the operation.
The expectations and method of postoperative bladder drainage should be discussed and determined prior to surgery. Patients should have no dietary restrictions and can resume light activities immediately. It is typically recommended that patients avoid heavy lifting and strenuous exercise for two months and sexual intercourse for about one month.
Some patients who have non-strenuous employment may be able to go back to work between one and two weeks, but it is generally advised not to plan on an early return to work or to schedule any important activities in the first two weeks. Patients may have significant fatigue or discomfort that might interfere with such plans.
In most cases, a patient who has only a simple incontinence operation will feel “back to normal” at two to three weeks, whereas a patient who has a major prolapse repair along with incontinence surgery may take four weeks or more to regain full strength and stamina. An individual’s response to the stress of surgery is of course highly variable and a person may use their response to other surgical procedures as a guideline.
IS SURGERY FOR ME?
The decision to have surgery to treat stress urinary incontinence is often a difficult one. When there are other symptomatic problems such as a prolapse or a significant gynecological disorder, then the choice of surgery becomes easier since these can be corrected at the same time.
The patient who has only SUI without significant urgency is more likely to be completely satisfied with the results of surgery, but mixed urinary incontinence is not a reason not to undergo surgery.
Many patients with mixed urinary incontinence will not have responded at all to medication for overactive bladder but may become completely dry after successful surgery for the stress leakage.
It all comes down to this: any patient in reasonable health for whom SUI is a significant social problem should seriously consider surgical treatment. The patient should also evaluate the non-surgical alternatives, and it is appropriate to spend a three-month period of time in a serious effort to strengthen the pelvic floor muscles prior to surgery.
There are a great number of different operative procedures for the treatment of stress urinary incontinence, and the patient should carefully discuss the alternatives with her physician.
Most importantly, the patient should feel comfortable asking her physician why a particular procedure is recommended and how much experience her doctor has in performing it. If treatment is not an option, women may also consider management options, such as absorbent products and urethral inserts.
FDA STATEMENTS REGARDING TRANSVAGINAL SURGICAL MESH
Six years ago the FDA issued (on October 20, 2008) a Public Health Notification (PHN) regarding potential complications associated with transvaginal placement of surgical mesh to treat pelvic organ prolapse (POP) and stress urinary incontinence (SUI). The PHN provided recommendations and encouraged physicians to seek specialized training in mesh procedures, to advise their patients about the risks associated with these procedures, and to be diligent in diagnosing and reporting complications.
In the six years since the FDA issued this PHN, the following has taken place:
On July 13, 2011, the FDA issued an update to the PHN in which it maintained that adverse events for POP mesh repair are not rare, as previously reported, and questioned the relative effectiveness of transvaginal mesh as a treatment for POP as compared to non-mesh surgical repair. Although the PHN made mention of mesh for use in SUI repair, the FDA did not address this SUI treatment approach and communicated that further evaluation would be required prior to releasing any statement in that regard.
The update continued to encourage physicians to seek specialized training in mesh procedures, to consider the risks associated with these procedures when making patient prescribing decisions, to advise their patients about these risks, and to be diligent in diagnosing and reporting complications.
On September 8 and 9, 2011, the FDA convened an Obstetrics and Gynecology Devices Panel of the Medical Devices Advisory Committee to further address the safety and effectiveness of transvaginal surgical mesh. The panel examined the use of transvaginal surgical mesh products to treat both pelvic organ prolapse (POP) and stress urinary incontinence (SUI).
At the hearings held on September 8 and 9, 2011, representatives of the TransvaginalMesh Device Manufacturers’ Working Group presented information to the FDA panel. This working group included representation by American Medical Systems, C.R. Bard, Boston Scientific, and Ethicon Women’s Health and Urology, in collaboration with the Advanced Medical Technology Association (AdvaMed) and Dr. Suzette Sutherland, a board-certified urologist focused in the area of female pelvic medicine and reconstructive surgery. We believe that this working group presented a very strong case for safety, efficacy, and continued classification of mesh as a Class II device to repair pelvic organ prolapse (POP) and stress urinary incontinence (SUI).
The recent FDA panel hearings in September 2011 supported recommendations by the agency that it reclassify transvaginal surgical mesh products for POP to Class III (pre-market approval) and require manufacturers of existing products to conduct additional post-market surveillance studies. Regarding surgical mesh devices for treatment of SUI, the panel supported the agency recommendation that such devices remain in Class II. Regarding retropubic and transobturator slings, the panel supported the working group position that no additional post-market surveillance studies are necessary. Regarding mini-slings, the panel recommended pre-market studies for new devices and additional post-market surveillance studies.
The FDA issued on January 3, 2012, requests for post-market surveillance studies of all previously approved and currently available vaginal mesh devices for pelvic organ prolapse (POP) and single-incision mini-sling devices with mesh for stress urinary incontinence (SUI). None of these devices were recalled and all remain available for usage in surgical procedures in women. These are all Class II 510K devices that had not required such data for their approval by the FDA. The FDA continues to assess whether to reclassify future vaginal mesh devices for POP from Class II to a stricter class III category. For more information, please visit the FDA’s update.
The FDA safety update is intended to inform patients and healthcare providers about complications associated with surgical mesh. NAFC has formed a position statement that you may read here: NAFC Position Statement on the Use of Vaginal Mesh in Pelvic Surgery.
Organizations presented information to an FDA advisory panel underlining case-specific details to patients with pelvic floor disorders. AUGS has also created a statement for patients who have undergone treatment using mesh or may be candidates for treatment. To view the AUGS/ACOG resources, please visit:
AUGS and ACOG Joint Committee Opinion on Vaginal Placement of Synthetic Mesh for POP (11/21/2011)
In January of 2016, following reports of thousands of injuries related to the devices, the FDA reclassified the devices from Class II, generally considered to be a moderate-risk, to Class III, considered high-risk. Additionally, the agency began requiring vaginal mesh manufacturers to submit “a premarket approval (PMA) application to support the safety and effectiveness of surgical mesh for the transvaginal repair of” pelvic organ prolapse.
Committee Opinion #513 “Vaginal Placement of Synthetic Mesh for Pelvic Organ Placement” is published in the December 2011 issue of Obstetrics & Gynecology. (12/2011)
RELATED ARTICLES

How Aging Changes Bladder Control — What Most People Don’t Expect
As an RN who’s worked in geriatrics for over 14 years, I know first hand that changes in bladder control can be a common part of aging. However, what I’ve also learned is that the treatment and management of these changes differ depending on whether they are due to aging or other conditions.
If you’re experiencing more frequent trips to the bathroom or sudden, intense urges to urinate, you might be wondering what is going on and what you can do about it. Let’s talk through some of the changes that might be going on in your body and how you can be better prepared to manage your bladder control.

Why Your Incontinence Product May Not Be Working Like It Used To
Most people assume that when an incontinence product leaks, the product itself failed. In reality, what often changes first is everything around it. Bladder leakage is not static. The amount of urine, how quickly it’s released, when leaks happen, and even how the body moves during sleep can all shift over time. A product that worked well six months ago may suddenly struggle under conditions it was never designed to handle.
This is especially common with overnight leakage, changes in mobility, medication adjustments, or shifts in bladder control related to aging and health conditions.
One of the biggest misconceptions is that absorbency alone determines performance. In reality, fit, absorption speed, body position, and wear duration all influence whether a product holds up in real-world situations.
Why So Many Women Put Themselves Last — and Why It Matters
Women are often the caregivers, planners, problem-solvers, and emotional anchors for everyone around them. They manage households, careers, children, aging parents, relationships, schedules, and responsibilities that rarely stop.
And somewhere in the middle of taking care of everyone else, many women quietly stop taking care of themselves.
They ignore symptoms. Delay appointments. Push through exhaustion. Normalize discomfort. Tell themselves they’ll deal with it “later.”
But later can turn into years.
This Women’s Health Month, it’s worth asking:
When was the last time you truly prioritized your own health

How Aging Changes Bladder Control — What Most People Don’t Expect
As an RN who’s worked in geriatrics for over 14 years, I know first hand that changes in bladder control can be a common part of aging. However, what I’ve also learned is that the treatment and management of these changes differ depending on whether they are due to aging or other conditions.
If you’re experiencing more frequent trips to the bathroom or sudden, intense urges to urinate, you might be wondering what is going on and what you can do about it. Let’s talk through some of the changes that might be going on in your body and how you can be better prepared to manage your bladder control.